VIDEO
This story is featured in today's TimesFreePress newscast.
Chattem officials say they're close to taking on what may be one of its biggest new products in the company's 134-year history.
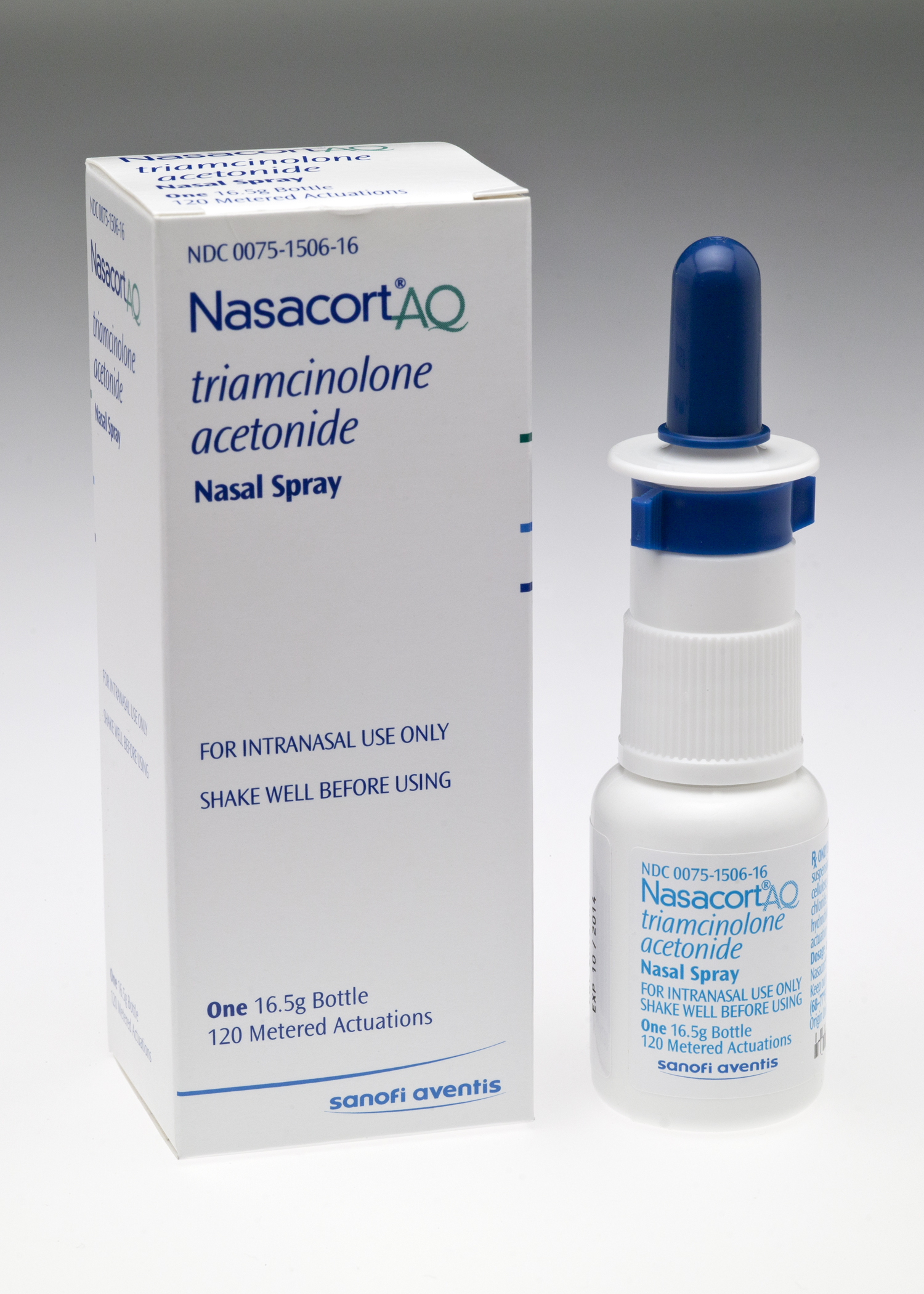
A U.S. Food and Drug Administration panel has recommended approval of Nasacort AQ Nasal Spray for over-the-counter use. If OK'ed by the FDA later this year, Chattem will oversee the shift of the current prescription medicine to in-store sales, and the product could over time become one of the Chattanooga-based company's biggest sellers behind allergy aid Allegra.
"It's a huge, huge market," said Chattem Chief Executive Officer Zan Guerry. "It will be significant."
The move would be expected to add more jobs in Chattanooga as Chattem would handle the packaging of Nasacort in the city, Guerry said. However, he wasn't sure yet how many employees could be added to the company's existing workforce of close to 600 in Chattanooga.
Nasacort AQ would be the first over-the-counter medicine in its class, according to Chattem parent Sanofi, the French drug giant. Nasacort would be aimed at providing temporary relief of nasal symptoms of hay fever or other upper respiratory allergies in adults and children 2 years of age and older.
"The OTC availability of Nasacort would continue to build on Chattem's highly successful OTC launch of Allegra and further expand our consumer health care offering," said Anne Whitaker, president of Sanofi's North America pharmaceuticals division, in a statement.
Guerry said that in the U.S. there are 50 million prescriptions a year for sprays such as Nasacort. That puts the category's sales at about $2 billion a year, he said.
"That gets your attention," he said. "It's a big opportunity."
Blair Ramey, Chattem's senior vice president of corporate development and media, said officials are upbeat about the potential switch of Nascort to over-the-counter use.
"If all goes well, the opportunity [to sell Nasacort on store shelves] is pretty exciting," he said.
Nasacort is made in England by Paris-based Sanofi.
FALL RELAUNCH
Meanwhile, this fall, Guerry said Chattem is looking to relaunch antacid Rolaids to the marketplace.
A new product for the first time ever will be a Rolaids liquid, he said, in addition to the tablets. Guerry said the rollout is generating a lot of buzz in the industry and Chattem expects to see more store displays and merchandising than officials had originally foreseen.
Earlier this year, Chattem bought Rolaids with plans to breathe new life into the product. The stomach med was pulled from shelves by former owner McNeil shortly after two 2010 recalls involving manufacturing problems.
Officials said Rolaids will go head-to-head with Tums, which they put as a $200 million-a-year brand.
Guerry said 2013 is shaping up as a solid year for Chattem overall. In addition to Allegra, which is Chattem's No. 1 product by sales, its Gold Bond lotions and powders have performed well, the CEO said.
The brand's lotion business is up 15 percent to 20 percent in sales at retail, he said. Gold Bond powders are 70 percent to 80 percent higher this year, Guerry said.
"We're having a very good year," he said. "Most of our brands are up."
Contact Mike Pare at mpare@timesfreepress.com or 423-757-6318.