VIDEO
This story is featured in today's TimesFreePress newscast.
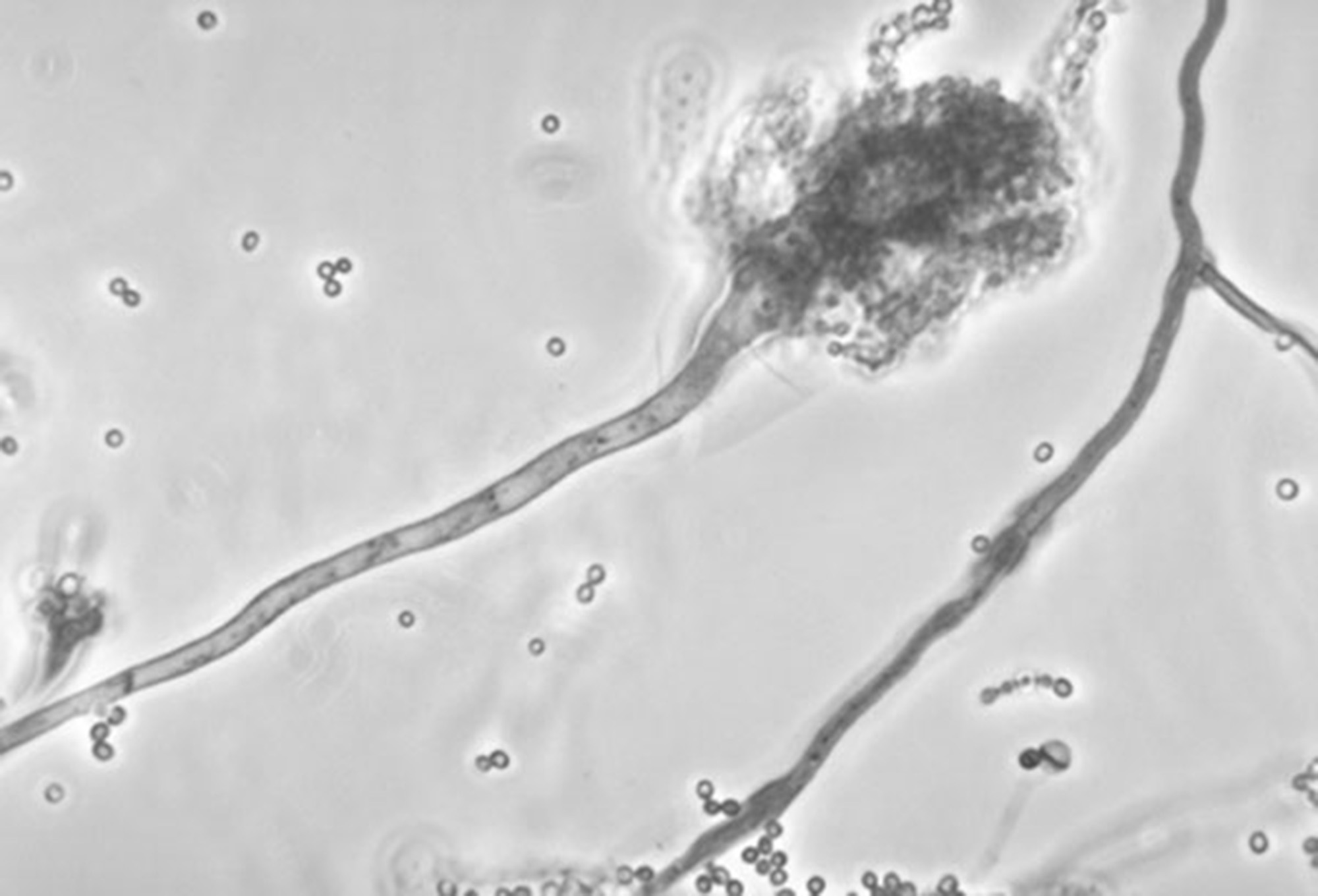 This undated photo made available by the Centers for Disease Control and Prevention shows a branch of the fungus Aspergillus fumigatus. The fungus can also cause skin infections if it enters a break in the skin. The meningitis outbreak is linked to the fungus being accidentally injected into people as a contaminant in steroid treatments. It's not clear how the fungus got into the medicine.
This undated photo made available by the Centers for Disease Control and Prevention shows a branch of the fungus Aspergillus fumigatus. The fungus can also cause skin infections if it enters a break in the skin. The meningitis outbreak is linked to the fungus being accidentally injected into people as a contaminant in steroid treatments. It's not clear how the fungus got into the medicine.Two Chattanooga health care providers are among dozens of hospitals and clinics served subpoenas this week in connection with a federal lawsuit against the drug manufacturer at the root of last year's devastating meningitis outbreak.
Erlanger Health Care System and Chattanooga Neurosurgery and Spine each were issued subpoenas Monday, as lawyers said that two patients -- one treated at each facility -- became sick from fungus-infested drugs that originated from the New England Compounding Center.
The subpoenas are the first public documentation linking Chattanooga-area patients to the drug-contamination crisis that sickened hundreds across the country.
The Centers for Disease Control and Prevention reports that more than 700 patients nationwide have confirmed illnesses related to tainted NECC product, and more than 240 have confirmed cases of meningitis.
At least 58 people have died after exposure to the drug -- 15 of whom were from Tennessee.
Last October, the Tennessee Department of Health identified Erlanger and 11 other area hospitals and medical facilities as providers that had received drugs from NECC.
At the time, Erlanger said the particular medications had not caused any known infections, that all NECC medications had been removed, and that potentially-affected patients had been notified.
In the subpoena, attorneys say they believe both Erlanger and Chattanooga Neurosurgery "purchased and received preservative-free methylprednisolone acetate from at least one of the three contaminated lots distributed by NECC."
There are more potentially contaminated products than just the methylprednisolone acetate (MPA) that was linked to meningitis. Other NECC drugs used to treat Tennesseeans include cardioplegic solution, used in heart surgeries, and eye solution.
The two Chattanooga-area victims included in the lawsuit are not identified in the subpoena, and attorneys would not confirm whether those patients developed meningitis or other illnesses.
The subpoenas request that the clinics produce documents that show all communications with and about NECC, including purchasing decisions, items bought, storage and pricing.
"We want to know what drugs they received from NECC, which were dispensed, how they were disposed of if they were not dispensed, and which patients received them. We want to know who ordered the drugs from NECC, and how they came in contact with them," said Gerard Stranch, a Nashville attorney representing some of the victims. Erlanger acknowledged it received the subpoena and is reviewing the lawsuit. Chattanooga Surgery and Spine did not respond to requests for a comment on Tuesday.
A statement from Stranch's law firm said that the "issuance of the subpoena should not be interpreted as an allegation of wrongdoing on the part of the clinic."
Clinics and doctors across the nation are being served similar subpoenas, as plaintiffs' attorneys trace how clinics distributed the tainted drugs.
The purpose of the subpoenas is "two-pronged," said Stranch. First, attorneys are trying to identify all potential victims who may not even know they're claimants in the case.
One of Stranch's Nashville clients was sent notice that he had been treated with drugs from the pharmacy. As a heart surgery patient, he didn't display any of the meningitis symptoms the hospital warned about. Instead, he developed septic wounds and abscesses that Stranch said were a result of contamination.
"He's not on the CDC website as a victim. He's not listed as someone who has meningitis or is related to that," said Stranch. "There very well could be 200 people in Tennessee who have some sort of complication as a result of receiving some kind of NECC-tainted product."
He said his firm is still getting calls from people who worry they may have contracted problems from the drugs.
Secondly, attorneys are trying to find out "exactly where these drugs went," Stranch said. The list of affected facilities compiled by the federal government last year is not all-inclusive, he said.
There are also questions as to whether Tennessee hospitals ordered the NECC drugs according to Tennessee law, which stated at the time that a prescription was needed for every dose of medication.
The NECC has been shut down and is in bankruptcy court.
Most of the cases against NECC have been consolidated in a single case now pending before a federal judge in Boston.
Stranch, who has been pursuing legal action on behalf of sickened patients since October, is one of the attorneys coordinating what's called "multi-district litigation."
He said the cases would likely be trial-ready in 2014.
All patient records released by the hospital in accordance with the subpoenas will be protected according to federal privacy laws, Stranch said.
Contact staff writer Kate Harrison at kharrison@timesfreepress.com or 423-757-6673.

